A Practical Guide to Vacuum-Assisted Headspace SPME For Routine Analysts and Non-Experts
What is Vacuum-Assisted HS-SPME and Why Should You Care?
If you’ve ever worked with headspace solid-phase microextraction (HS-SPME), you know the trade-off: volatile compounds extract quickly, but semi-volatiles can take forever to reach equilibrium. The usual solution? Heat the sample. But heating comes with problems—thermally labile compounds decompose, artifacts form, and you might end up with an extract that doesn’t truly represent your original sample.
Vacuum-assisted HS-SPME (Vac-HS-SPME) offers a different path. Simply put: you evacuate the air from the sample container before extraction. That’s it. One extra step that can dramatically speed up extractions or let you work at much lower temperatures.
This guide explains what happens when you pull a vacuum, why it works for some compounds but not others, and how to use it in your daily work—without getting lost in complex equations.
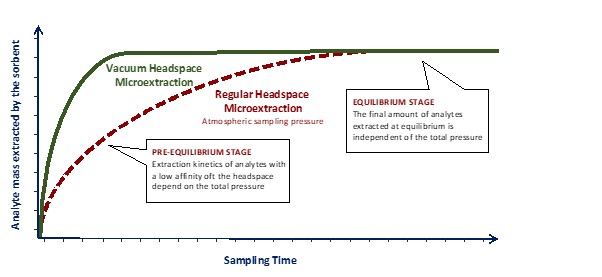
Think about what happens during a normal headspace extraction:
- Analytes must evaporate from the sample into the headspace
- They must travel through the air to reach the fiber
- The fiber coating takes them up
For volatile compounds, this happens quickly. But for semi-volatiles, evaporation is slow—the headspace becomes a bottleneck.
When you pull a vacuum, you remove most of the air molecules from the headspace. With fewer air molecules in the way, analyte molecules can diffuse (move through the gas phase) much faster—about 25 times faster when you go from atmospheric pressure down to typical vacuum levels used in this technique.
The key insight: You’re not changing how much analyte the fiber can ultimately extract at equilibrium—that amount stays the same. You’re just helping it get there faster.
Here’s the practical question: When should you bother with vacuum?
For water and water-containing samples, the answer lies in a property called the Henry’s Law constant (KH) . Don’t let the name intimidate you—it’s simply a measure of how readily a compound moves from water (or a water-containing sample) into air.
– High KH compounds (volatiles): They evaporate easily. The bottleneck is in the liquid phase, not the gas phase. Vacuum won’t speed them up much.
– Low KH compounds (semi-volatiles): They struggle to evaporate. The gas phase is the bottleneck. Vacuum helps them significantly.
The rule of thumb: If your compound has a KH value ≤ 1.6 × 10⁻⁴ atm m³ mol⁻¹, vacuum will accelerate its extraction. Above this threshold, liquid-phase limitations dominate, and vacuum won’t make much difference.
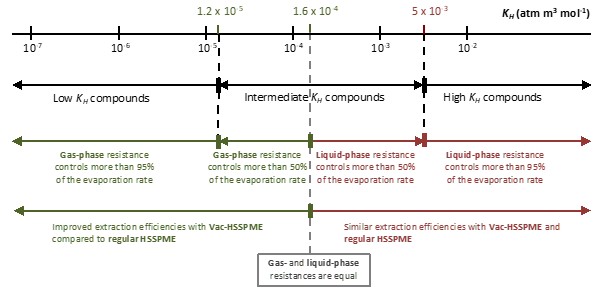
*Example:* Polycyclic aromatic hydrocarbons (PAHs) like Pyrene benefit from vacuum. Napthalene? Not so much—it’s volatile enough already.
About Solid Samples?
For solids, the situation is similar but slightly more complicated. When you apply vacuum:
– Analyte diffusion in the gas phase speeds up (same as with liquids)
– This increases the vapor flux at the solid surface
– Extraction proceeds faster
Many analysts add a small amount of water to solid samples before Vac-HS-SPME. Why? The water helps release analytes from the solid matrix and then vacuum accelerates their evaporation from that water layer. It’s a one-two punch.
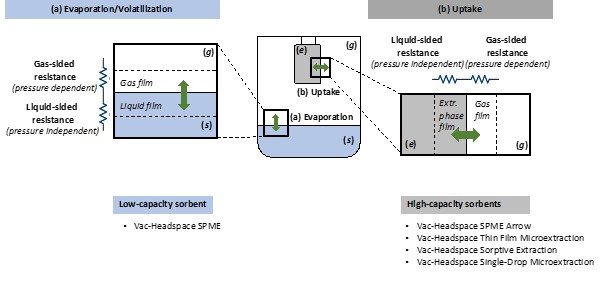
Here’s something interesting that researchers discovered: sometimes the fiber uptake itself can be the slow step—especially with thin-film microextraction (TFME) devices that have larger extraction volumes.
When a compound has a “high affinity for the extraction phase” (meaning it really likes to get into the coating), it takes longer to reach equilibrium between headspace and fiber. Vacuum helps here too, by speeding up how fast analyte molecules can reach the fiber surface.
This means that even for some compounds that evaporate readily, vacuum might still help if their uptake by the fiber is gas-phase limited.
- Faster Extractions
The most obvious benefit. Compounds that used to take 60-90 minutes to reach equilibrium might need only 30-40 minutes under vacuum. In some published methods, extraction times were cut in half or better. - Lower Temperatures (or Even No Heating)
This is where Vac-HS-SPME really shines. Because extraction is faster, you don’t need to crank up the heat to drive analytes into the headspace. In fact with vacuum you can even extract your sample at temperatures as low as 5 oC, which is ideal for the analysis of fish, dairy products.
Real-world examples:- Cannabinoids in hemp: Vac-HS-SPME at 90°C vs. regular HS-SPME at 150°C—no thermal decomposition of CBD, no artifacts
- Fish volatiles: Vac-HS-SPME at 5°C gave comparable results to regular HS-SPME at 30-40°C
- Wine haloanisoles: 30 min at 25°C under vacuum outperformed 30 min at 55°C at atmospheric pressure
- Preserving Sample Integrity Lower temperatures mean:
- No thermal degradation of labile compounds
- Fewer artifacts from sample heating
- A more representative “snapshot” of what’s actually in your sample
- Better Sensitivity in Less Time
Because you’re reaching equilibrium faster (or getting closer to it in a given time), you often end up with higher extracted amounts within your chosen extraction time.
You might think: if vacuum helps and heating helps, wouldn’t combining them be even better?
Sometimes yes, sometimes no. It depends on your sample and your fiber.
The Surprising Finding
For standard SPME fibers (especially PDMS), heating during vacuum extraction sometimes “reduced” performance. Why? Water vapor. When you heat a water-containing sample under vacuum, you generate a lot of water vapor that can compete with analytes for the fiber coating. But, with vacuum only mild heating is needed so, you might not never record this effect.
Besides, with SPME Arrow and TFME devices that have larger PDMS volumes, heating under vacuum worked beautifully. The large PDMS volume could handle the water competition while still benefiting from both vacuum and temperature.
For Viscous Samples (Like Olive Oil)
Here, mild heating during vacuum extraction is actually essential. Viscous samples have liquid-phase limitations—analytes move slowly through the oil to reach the surface. A little warmth (e.g., 43°C) reduces viscosity by ~40% and removes that bottleneck, while vacuum handles the gas-phase transport.
The Basic Workflow
- Place your sample in a gas-tight container designed for vacuum use
- Evacuate the air using a vacuum pump (or manually with a syringe/piston)
- Introduce the SPME fiber through a septum or valve
- Extract for your chosen time (typically shorter than your regular method)
- Analyze as usual
Critical Practical Points
- Evacuation timing matters
- For liquid samples: You can evacuate before or after introducing the sample
- For solid samples: You typically evacuate with the (frozen) sample present.
Warning: Volatile compounds can be lost during evacuation if you’re not careful. Keep evacuation times short, or freeze the sample first to reduce headspace concentrations of volatiles.
-Vacuum level: Typical target is around 0.04 atm (about 30 mm Hg). A good vacuum pump is recommended over manual methods for consistent results.
Common Questions from Analysts
Q: Which Fibers Work?
A: All common SPME coatings have been used successfully:
Q: Will I lose volatiles during air evacuation?
A: Possibly, if you’re not careful. Keep evacuation time short (a few seconds to a minute, depending on your setup). If very volatile analytes are present, freeze the sample before evacuation—this minimizes their presence in the headspace during pumping.
Q: Do I need to re-optimize my entire method?
A: Yes, but it’s straightforward. Start with your regular method parameters, then explore shorter extraction times and/or lower temperatures. The published literature for your analyte type can give you good starting points.
Q: What about matrix effects—ethanol, salts, pH?
A: These affect partitioning just as they do in regular HS-SPME. The vacuum effect is additive. In wine (with ethanol), vacuum still provided benefits despite the higher total pressure from volatile matrix components.
Q: Can I automate this?
A: Vial closures that allow vacuum evacuation and sampling within autosampler workflows. This technology is emerging and should become more accessible.
Good candidates for vacuum:
- Semi-volatile compounds
- Thermally labile compounds that decompose with heating
- Samples where you want to preserve the true volatile profile
- Perishable samples (like fish) where low-temperature extraction is beneficial
- Viscous samples (with mild heating)
Less benefit:
- Highly volatile compounds that already extract quickly
- Samples where liquid-phase kinetics are overwhelmingly slow (though mild heating can help)
The Bottom Line
Vacuum-assisted HS-SPME isn’t complicated. It’s regular HS-SPME with one extra step: pulling the air out before you start. That simple change can:
- Cut extraction times dramatically
- Lower your sampling temperatures
- Preserve sample integrity
- Give you more representative results
The fundamentals are well understood. The practical benefits are proven across many applications—water, soil, food, wine, biological samples. The only thing holding it back from wider adoption has been the lack of commercial off-the-shelf hardware, but that’s changing.
If you’re struggling with long extraction times or thermal degradation of your targets, vacuum assistance is worth exploring. Start with the published methods for your analyte type, get or make a suitable vacuum vessel, and see what it can do for your work.
